 In his honor, element 101 was named after him, mendelevium. The periodic table was invented by the Russian chemist Dmitry Ivanovich Mendeleyev (1834–1907). There are also many other patterns and relationships. caesium is much more metallic than helium). For example, elements to the bottom and far left of the table are the most metallic, and elements on the top right are the least metallic. There are 3 main groups in the Periodic Table metals, metalloids, and nonmetals. The periodic table has been used by chemists to observe patterns and relationships between elements. In 1990 the International Union of Pure and Applied Chemistry (IUPAC) decided to use the new system with Arabic numerals, to replace the two old group systems that used Roman numerals. The Roman numeral names were used in most of the 20th century. There are two systems of group numbers one using Arabic numerals (1,2,3) and the other using Roman numerals (I, II, III). For example, group 18 is known as the noble gases because they are all gases and they do not combine with other atoms. Elements in a group have electrons arranged in similar ways, according to the number of valency electrons, which gives them similar chemical properties (they behave in similar ways). There are 18 groups in the standard periodic table. Elements in a period have consecutive atomic numbers.Ī column of elements down the table is called a group. 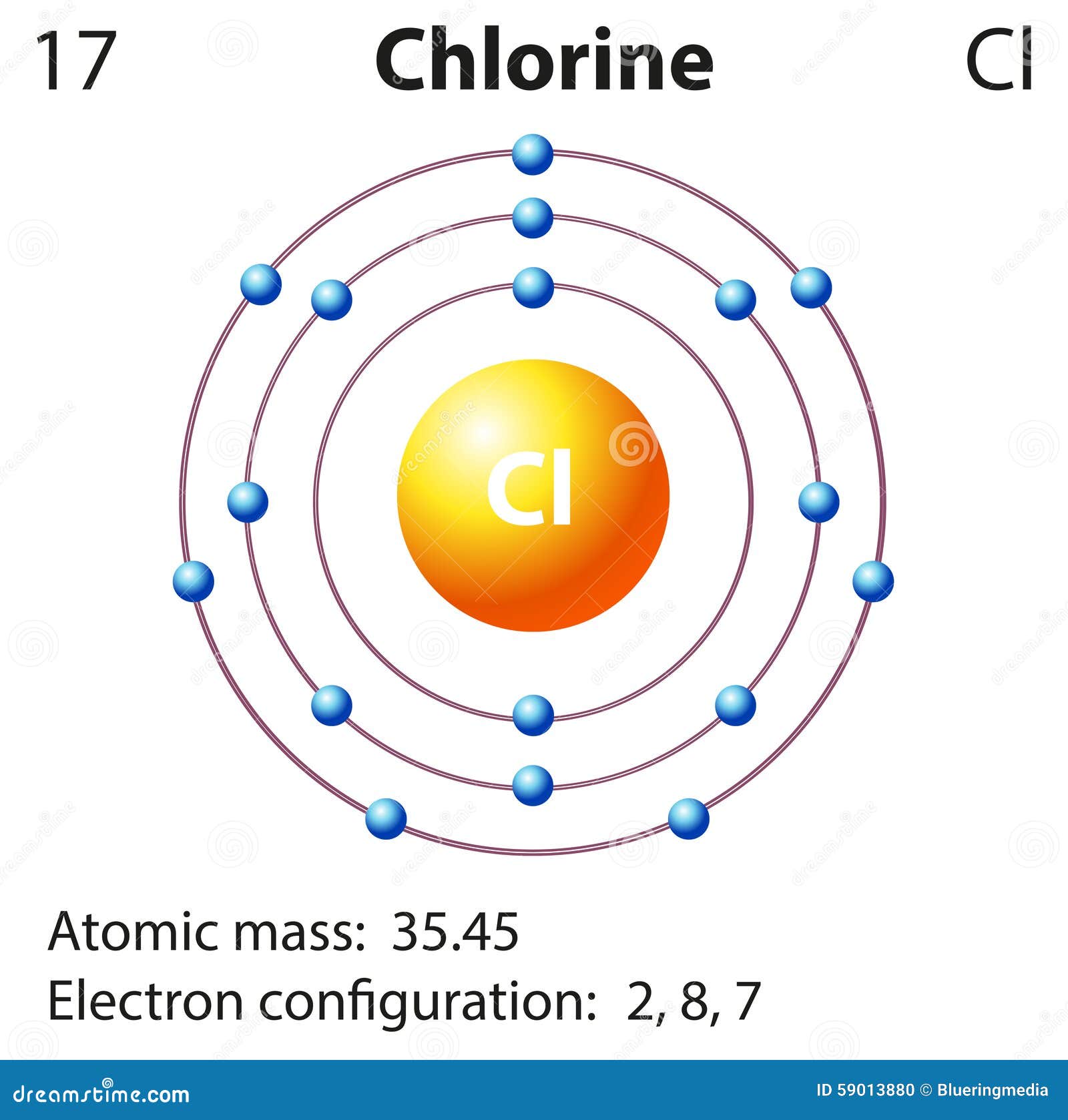
Period 2 and Period 3 both have 8 elements. Period 1 has only 2 elements in it: hydrogen and helium. Toxic as a gas, but in form of chloride ions. Chemical element and halogen with symbol Cl and atomic number 17. A row of elements across the table is called a period. Chlorine on periodic table of the elements. In the periodic table the elements are arranged into periods and groups. The atomic number of an element is the same as the number of protons in that particular nucleus of an atom. However, the Periodic table generally displays only the symbol of the element and not its entire name. In the table, the elements are placed in the order of their atomic numbers starting with the lowest number of one, hydrogen. The periodic table of elements is widely used in the field of Chemistry to look up chemical elements as they are arranged in a manner that displays periodic trends in the chemical properties of the elements. Elements that have similar characteristics are often put near each other. 
The periodic table is a table that puts all known chemical elements in a specific order. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
